Fire in the Sky Energy Science-SMART! Home
Science Concept: What Is Static Electricity?
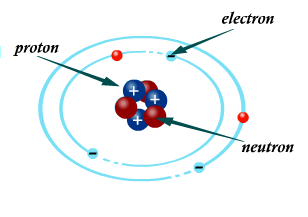
Atoms contain charged particles called protons and electrons. Protons have a positive charge, while electrons have a negative charge. When an atom has the same number of protons as electrons, the charges cancel each other out and the atom is electrically neutral.
But atoms can lose or gain electrons. An atom that loses electrons becomes positively charged. An atom that gains electrons becomes negatively charged. The buildup of electric charge (either positive or negative) on an object is called static electricity. (The word static means “not moving.”)
Static electricity is different from electrical current in that it does not flow continuously. Electrons move from one atom to the next creating a flow or current. In static electricity, the outer electrons from one substance get free from their atoms and can attach to another substance, thus giving the second substance a negative charge. Electrons can get free when two items rub together—like your shoes rubbing across the rug.
Static electricity is the imbalance of positive and negative charges. Two things with opposite charges will pull towards each other.





